Options for treatment of renal cell carcinoma are multiplying. Patients are benefiting from rising survival rates but also must be managed to minimize acute kidney injury from cancer treatment, with nephrectomy the greatest risk in RCC. That means more teamwork between specialties at the VA and elsewhere.
ANN ARBOR, MI—The rapidly multiplying options for treatment of renal cell carcinoma, their range of side effects and rising survival rates have increased the need for teamwork between oncologists and nephrologists.
Acute kidney injury occurs in up to 30% of patients with cancer. Renal cell carcinoma, which accounts for about 70% to 80% of all kidney cancer, poses a particular risk of kidney injury.
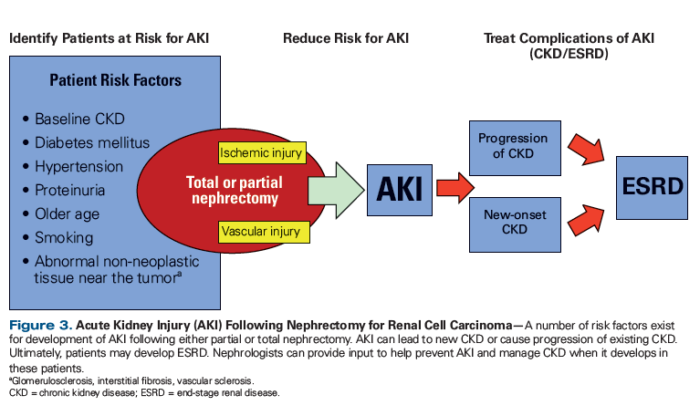
Nephrectomy is associated with a more than 10% risk of ischemic or vascular kidney injury, although partial nephrectomy reduces that risk and is now recommended whenever feasible, said Mark A. Perazella, MD, an oncologist at the VAMC in West Haven, Conn., professor of medicine at the Yale University School of Medicine and director of the Acute Dialysis Program at Yale-New Haven Hospital and his co-author Mitchell Rosner, MD, chair of the University of Virginia Department of Medicine.
Chemotherapy, targeted therapies and immunotherapies for renal cell carcinoma also contribute to the occurrence of drug-induced acute and chronic kidney injury in cancer patients, according to an article published last year in Oncology.1
Nephrologists, working with oncologists and urologists, can play a vital role in managing patients with chronic kidney disease who develop RCC and patients with RCC who develop acute kidney injury or chronic kidney disease as a result of treatment, according to Perazella and his colleagues.
“When kidney disease develops in the setting of RCC, mortality is significantly increased with patients often dying of a non-cancer-related complication of kidney disease,” they wrote in a review published last fall in Kidney International. “Nephrologists should be involved in all aspects of the care of patients with RCC including in the acute setting prior to nephrectomy and in the chronic setting for patients with post-nephrectomy chronic kidney disease and those receiving potentially nephrotoxic anti-cancer agents.” 2
The involvement of nephrologists may be particularly important at the VA, which has high rates of both RCC and chronic kidney disease.
Nationally, the number of patients diagnosed with renal cell carcinoma has steadily increased. According to the U.S. Centers for Disease Control and Prevention, the incidence rate per 100,000 people for RCC rose from 12.2 in 1999 to 16.6 in 2015.
The American Cancer Society estimates that about 44,120 men and 29,700 women will receive a diagnosis of kidney cancer this year and more than 50,000 of them will have RCC. Of those, about 50,000 will have renal cell carcinoma. Veterans will account for more than 4% of RCC diagnoses.
RCC is far more common among veterans because they have a higher rate of known risk factors such as obesity, smoking, diabetes and hypertension. Those factors drive the incidence rate up to 38.1 per 100,000 in veterans, more than twice that seen in the overall population.
Chronic kidney disease also disproportionately affects veterans. Researchers at the VA found that nearly half of veterans who had an outpatient visit at any time during 2007 had chronic kidney disease.3 Nationally, about 15% of the adult population has CKD, though 90% are undiagnosed, according to the National Kidney Foundation.
Recent Approvals
The current pace of U.S. Food and Drug Administration approvals makes keeping track of the recommended therapies for metastatic RCC and their effects on the kidneys a significant challenge for oncologists and nephrologists.
“The current standard for treatment of clear-cell renal cell carcinoma is changing every three months or so,” explained Ajjai S. Alva, MBBS, an oncologist with the VA Ann Arbor Healthcare System and an associate professor of medical oncology at the University of Michigan Medical School.
The FDA approved cabozantinib monotherapy in 2016 for advanced RCC after anti-angiogenic therapy. In 2017, cabozantinib received approval for use as a first-line therapy for advanced RCC based on the Phase II CABOSUN trial, which showed a 52% reduction in the risk of disease progression or death and a 62% increase in median progression-free survival. Like the other recent studies, CABOSUN compared the new agent to sunitinib, which had been the standard of care for first-line treatment of metastatic RCC. Cabozantinib is a preferred therapy for poor/intermediate risk patients, according to National Comprehensive Cancer Network guidelines.
“Cabozantinib blocks a broader spectrum of proteins, including c-Met and VEGFR2 as well as AXL and RET,” Alva told U.S. Medicine. “That’s better than sunitinib.”
Lenvatinib plus everolimus also received approval in 2016 for patients with advanced RCC following antiangiogeneic therapy on the strength of the Phase II Study 205. That trial showed a 63% reduction in the risk of death or progression for the combination compared to everolimus alone and better response rate, progression free survival and overall survival compared to lenvatinib alone.
In April 2018, the combination of nivolumab and ipilimumab gained approval for treatment naïve intermediate or poor-risk patients, following publication of the results of the CheckMate 214 trial that showed a 37% improvement in overall survival, a significant increase in response rate, including a 9% complete response rate and extended progression-free survival.
The FDA granted pembrolizumab monotherapy approval in the first line after the KEYNOTE 427 trial showed a 38.2% response rate.
Pembrolizumab plus axitinib gained approval in March 2019 based on the KEYNOTE 426 trial, which found the combination had an objective response rate of 59.3% and increased 12-month and 18-month overall survival, 89.9% vs. 78.3% and 82.3% vs. 72.1%, respectively, compared to sunitinib. When reported, median overall survival had not been reached in either arm, but results indicated an at least 47% improvement for the new combination.
“The study shows that an immunotherapy on top of a tyrosine kinase inhibitor (TKI) is better than a TKI alone,” Alva said. “Patients live longer across all prognostic risk groups; they go longer before they need to change treatment and demonstrate response using RECIST measures—all three co-primary end points.”
The JAVELIN Renal 101 trial showed that avelumab plus axitinib extended progression-free survival and doubled response rates across risk groups when compared to sunitinib. The FDA granted the combo priority review in February 2019.
“This combination is slightly different in that it goes for a programmed death ligand rather than the receptor, though most researchers think the two approaches have about the same efficacy. Again, it showed that two are better than just sunitinib,” Alva said.
Lenvatinib plus pembrolizumab was granted breakthrough therapy designation based on a Phase Ib/II study which showed objective response rates of 83% in treatment naïve patients and 50% in those who had been previously treated. A Phase III trial is in progress.
More Therapies, More Questions
“We now have three combinations with roughly the same efficacy,” Alva noted. “So now we need to determine, which patients for which therapies? What markers can guide treatment?”
Because kidney cancer is characterized by relatively few mutations, unlike lung cancer or bladder cancer, it’s always been an outlier, Alva said.
But researchers have thought for some time that there are “at least two sets of patients in RCC, one driven by VEGFR [vascular endothelial growth factor receptor] and the other immune system driven, and that’s exactly what we’re seeing. These combinations take the guesswork out by covering both baskets, but toxicity is higher,” he explained.
To manage the toxicity and really improve outcomes, “better biomarkers are needed,” Alva stressed. “How many drugs can you combine and patients take? There’s a ceiling to that. Weaker patients may choose to take just one and add the other if they are doing well functionally.”
1 Perazella MA, Rosner MH. Acute Kidney Injury in Patients with Cancer. Oncology. 2018 July 15;32(7):351-9.
2 Perazella MA. Renal cell carcinoma for the nephrologist. Kidney Int. 2018 Sep;94(3):471-483.
3 Patel N, Golzy M, Nainani N, Nader ND, Carter RL, Lohr JW, Arora P. Prevalence of various comorbidities among veterans with chronic kidney disease and its comparison with other datasets. Ren Fail. 2016;38(2):204-8.
