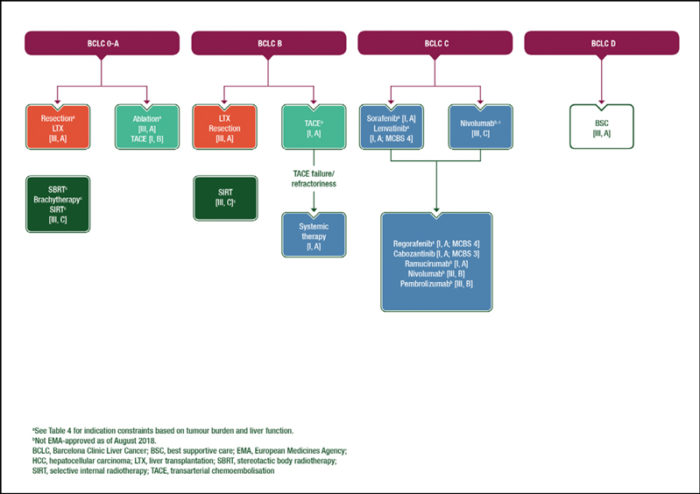
RICHMOND, VA—For more than a decade after the U.S. Food and Drug Administration approved sorafenib for hepatocellular cancer, treatment options for the aggressive malignancy remained static. That changed dramatically in the last 24 months as five drugs received approval for HCC—nivolumab, regorafenib, pembrolizumab, lenvatinib and cabozantinib.
Effective treatment of HCC is often stymied by late diagnosis, advanced disease and other complications. The VA estimates that fewer than “50% of patients with HCC undergo definitive treatment because of age, liver function, general medical condition, and patient refusal.”
Transplantation is widely recognized as the treatment most likely to offer a cure for HCC, but relatively few patients meet the Milan guidelines for suitable candidates. A chronic shortage of livers further restricts the number of patients who can benefit from transplantation. Resection may also be curative, though the VA viral hepatitis and liver disease website notes that “less than 20% of HCC patients are good candidates for surgical resection.”
For example, patients who aren’t candidates for surgery have localized treatment options, including transcatheter arterial chemoembolization, radiofrequency ablation, brachytherapy, percutaneous injection of ethanol or ascetic acid, and hepatic artery chemotherapy.
More Options
For patients with large or multiple tumors or metastases, however, the options have been quite limited. Until recently, sorafenib, which gained FDA approval for advanced HCC in 2007, has been the only systemic chemotherapy shown to extend survival for patients with unresectable HCC. The SHARP clinical trial showed sorafenib, an oral multikinase inhibitor, extended overall survival from 7.9 months to 10.7 months compared to no treatment, a 31% improvement. 1
Last August, lenvatinib received FDA approval for firstline use in unresectable HCC. Another multikinase inhibitor, lenvatinib showed noninferiority to sorafenib in the multicenter, global phase 3 REFLECT trial, which enrolled 1,492 treatmentnaïve patients. In that study, median overall survival was 13.6 months with lenvatinib vs. 12.3 months with sorafenib, indicating an 8% reduction in mortality risk for lenvatinib. Lenvatinib also significantly extended median progression-free survival to 7.4 months from 3.7 months, a 34% improvement. More patients responded to lenvatinib compared to sorafenib as well, 24.1% versus 9.2%. 2
“Sorafenib has a higher incidence of hand-foot syndrome (which is the most disabling side effect to treat) while lenvatinib has more hypertension, proteinuria and hypothyroidism. I personally find the latter easier to manage,” said Binu John, MD, MPH, associate professor of Medicine, Virginia Commonwealth University, and director of liver transplantation and the liver cancer program at Hunter Holmes McGuire VAMC, Richmond, VA.
Ann Oncol 2018; 29 (Suppl 4): iv238–iv255.
In choosing between the two, “some could argue that, if patients progress on sorafenib, they have multiple options, since all trials on regorafenib, cabozantinib and anti-PD1 [agents] were done in patients who progressed post-sorafenib or were intolerant. There is no such data with lenvatinib, since it just got approved,” John told U.S. Medicine.
The other drugs received approval for use in the second- or subsequent lines, which raises new questions about appropriate sequencing. “Regorafenib, nivolumab and cabozantinib are all options,” John said. “I think the enthusiasm for pembrolizumab may be diminished after the early release of the Phase 3 study, which was negative, although we have not seen it in print.”
Pembrolizumab received FDA approval in November 2018, but in February, its manufacturer reported that the KEYNOTE-240 trial failed to significantly extend survival compared to best supportive care plus placebo in previously treated patients with advanced HCC.
Regorafenib gained secondline approval in April 2017 based on the RESORCE trial, which enrolled 573 patients. That trial showed that regorafenib improved median progression free survival to 3.1 months from 1.5 months for placebo and overall survival to 10.6 months from 7.8 months. Grade 3 or 4 treatment-related adverse events that occurred at much higher rates in the regorafenib group than among the placebo group included hypertension (15% vs. 5%) and hand-foot skin reaction (13% vs. 1%). 3
The data on regorafenib “is impressive, but it is a drug that is difficult to manage,” John noted.
Nivolumab received accelerated approval in late 2017 for use after progression on or intolerance of sorafenib. The Phase 1/2 CheckMate-040 trial with 154 patients found that 14.3% of patients responded to nivolumab, and duration of response ranged from 3.2 months to more than 38.2 months. Among responders, 91% continued to respond for more than six months, and 55% responded for at least 12 months. 4
An ongoing head-to-head trial has stimulated interest in nivolumab. “We are awaiting the Phase 3 Checkmate-459 data comparing sorafenib and nivolumab,” John said. “If that is a positive study, nivolumab will become first line.”
Most recently, cabozantinib emerged as a second-line treatment option following soratenib. Approved in January 2019, cabozantinib is another multiple tyrosine kinase inhibitor. The Phase 3 CELESTIAL trial with 707 patients was stopped early, based on strong interim results. Cabozantinib demonstrated a 24% reduction in mortality risk and a median improvement in overall survival from 8.0 months to 10.2 months compared with placebo. 5
When combined with the improvement in median progression survival, 5.2 months vs. 1.9 months, cabozantinib demonstrated a 56% reduction in the risk of disease progression or death. Nearly two-thirds of patients in the cabozantinib group had either partial response or stable disease compared to one-third of patients receiving placebo.
1. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008 Jul 24;359(4):378-90.
2. Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, Baron A, Park JW, Han G,Jassem J, Blanc JF, Vogel A, Komov D, Evans TRJ, Lopez C, Dutcus C, Guo M, Saito K, Kraljevic S, Tamai T, Ren M, Cheng AL. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018 Mar 24;391(10126):1163-1173.
3. Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, Gerolami R, Masi G, Ross PJ, Song T, Bronowicki JP, Ollivier-Hourmand I, Kudo M, Cheng AL, Llovet JM, Finn RS, LeBerre MA, Baumhauer A, Meinhardt G, Han G; RESORCE Investigators. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebocontrolled, phase 3 trial. Lancet. 2017 Jan 7;389(10064):56-66.
4. El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Dela Cruz C, Lang L, Neely J, Tang H, Dastani HB, Melero I. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017 Jun 24;389(10088):2492-2502.
5. Abou-Alfa GK, Meyer T, Cheng AL, El-Khoueiry AB, Rimassa L, Ryoo BY, Cicin I, Merle P, Chen Y, Park JW, Blanc JF, Bolondi L, Klümpen HJ, Chan SL, Zagonel V, Pressiani T, Ryu MH, Venook AP, Hessel C, Borgman-Hagey AE, Schwab G, Kelley RK. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N Engl J Med. 2018 Jul 5;379(1):54-63.
